12 May “In vitro” bacteriostatic and antibacterial effect of DigestoCid© and two competing products

Fundamentals of the experimental design
This experiment uses a dilution-neutralization method that is based on the official European method EN 1040:2005, “Chemical disinfectants and antiseptics – Quantitative suspension test for the evaluation of basic bactericidal activity of chemical disinfectants and antiseptics”.
The fundamentals of the method are as follows:
To estimate the antimicrobial activity of a disinfectant or antiseptic, a dilution of the test compound is mixed with a bacterial cell suspension and, after a period of incubation, the capacity of the remaining bacteria to grow is measured.
In the case of weak acids, it has been established that their antimicrobial activity in vitro can be overestimated if a neutralizer is not applied. The reason of the overestimation is that weak acids act on bacteria through two mechanisms of action:
- Acidic stress, through a low environmental pH
- The diffusion of the undissociated weak acid through the bacterial cell membrane, followed by intracellular dissociation of the weak acid, that acidifies the cellular environment and the disrupts the bacterial metabolism.
Whenever a weak acid is used as an antiseptic or disinfectant, the antimicrobial action through lowering the environmental pH (Mechanism of action nº1) will vary depending to the conditions of application of the acid. In example, whenever the same amount of weak acids is applied to the drinking water of poultry farms, the final pH of the drinking water will depend on the initial pH and hardness of the water. The same happens when the weak acid travels through the digestive system of the animal because each digestive organ has a different pH.
For this reason, in vitro tests for weak acids as antiseptics focus on the mechanism nº2, the ability of the weak acid to penetrate the bacterial cell wall and disrupt its metabolism.
By adding a neutralizer, in this case the buffer in the peptone water and the disodium hydrogen phosphate, the antimicrobial activity through mechanism of action nº1 is stopped or greatly attenuated, and the in vitro test mainly measures the the antimicrobial activity through mechanism of action nº2.
Materials
- Buffered peptone water broth (BPW)
- Muller Hinton (MH) agar
- ATCC 14028 Salmonella strain
- ATCC 25923 E.coli strain
- Disodium hydrogen phosphate
- Products to test: DigestoCid©, Competing product A, Competing product B
- Incubation tubes and Petri Dishes
Procedure
- Dilute DigestoCid©, Competing product A, Competing product B to 5%.
- Prepare BPW. Dispense into 3*8*2 tubes, 8ml/tube.
- Add 8ml of each diluted product to the first test tube, then carry a gradient dilution. The final concentration of acidifier are as follows:

- Add 1ml of 0.015 mol / L disodium hydrogen phosphate to each test tube, mix well and let stand for 5min.
- Add 1 ml of 103 CFU/ml Salmonella suspension or 1ml 103 CFU/ ml E. coli suspension to each test tube.
- Incubate at 37°C for 24 hours.
- Minimum Inhibitory Concentration: After incubation, observe the presence of turbidity. The lowest concentration without turbidity is considered the Minimum Inhibitory Concentration. The Minimum Inhibitory Concentration (MIC) is defined as the lowest concentration of an antimicrobial ingredient or agent that is bacteriostatic (prevents the visible growth of bacteria).
- Minimum Bactericidal Concentration:Take 1 ml of the cultured solution into a Petri Dish and pour 15 ml MH agar. Incubate at 37 ° C for 48 hours. Observe the growth of colonies. The lowest concentration that doesn’t show growth of colonies is the Minimum Bactericidal Concentration. The Minimum Bactericidal Concentration (MBC) is the lowest concentration of an antibacterial agent required to kill a bacterium over a fixed, somewhat extended period under a specific set of conditions. It can be determined from the broth dilution of MIC tests by subculturing to agar plates that do not contain the test agent. The MBC is identified by determining the lowest concentration of antibacterial agent that reduces the viability of the initial bacterial inoculum by a pre-determined reduction such as ≥99.9%. The MBC is complementary to the MIC; whereas the MIC test demonstrates the lowest level of antimicrobial agent that greatly inhibits growth, the MBC demonstrates the lowest level of antimicrobial agent resulting in microbial death. In other words, if a MIC shows inhibition, plating the bacteria onto agar might still result in organism proliferation because the antimicrobial did not cause death. Antibacterial agents are usually regarded as bactericidal if the MBC is no more than four times the MIC.
Results
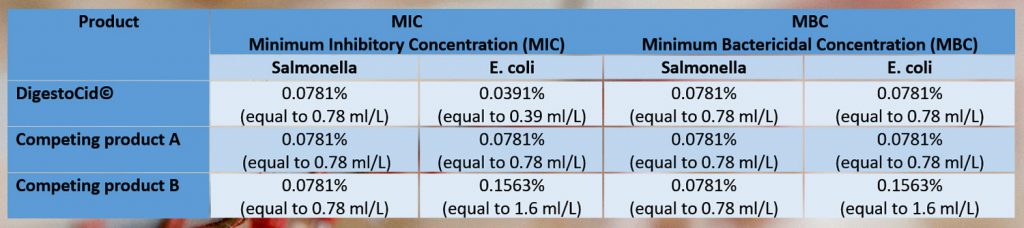
Recommended doses of the products
- DigestoCid©: 0.5-1 ml/L of drinking water
- Competing product A: 1.5-2 ml/L of drinking water
- Competing product B: 1-2 ml/L of drinking water
Conclusions
When determining the bacteriostatic effect of the three products (Minimum Inhibitory Concentration, MIC), we found that all the products had the same MIC against Salmonella, while DigestoCid© had the lowest MIC against E.coli, followed by Competing product A and Competing product B.
When determining the antibacterial effect of the three products (Minimum Bactericidal Concentration, MBC), we found that all the products had the same MBC against Salmonella, while DigestoCid© and Competing product A had the lowest MBC against E.coli.
It can be concluded that DigestoCid© is the most effective against Salmonella and E.coli “in vitro”.
This trial proves that DigestoCid© is effective as a water sanitizer at the recommended commercial doses (0.5-1 ml/L of drinking water) because its MIC and MBC for Salmonella and E.coli range between 0.39 ml/L and 0.78 ml/L.
Its lower recommended dose compared to competing products A and B is another competitive advantage of DigestoCid©.
Product of choice
DigestoCid© is indicated as an acidifier, sanitizer of drinking water and to improve intestinal health in poultry and pigs of all ages. It contains:
- Buffered organic acids, which reduce the pH of the water and inhibit the growth of Gram-negative bacteria.
- Monoester fatty acids, also called medium chain fatty acids, which inhibit the growth of Gram-negative and Gram-positive bacteria. In addition, they have antiviral effects.
- Essential oils and phytochemicals with antibacterial, antifungal, antiviral and antioxidant properties.
DigestoCid© has a broader spectrum of action than products based solely on organic acids, allowing superior antibacterial and antifungal efficacy at a less acidic pH (5.5-6.5).

Certain health statements may not be applicable in your region.

